Date: September, 2022 | Category: Company | Author: Hana Trokic
With live events back in full swing this year, GlobalVision is taking notes and taking full advantage.
The ideal opportunity to network and gain insights into new innovations in the industry, we’re excited to be face-to-face with some of pharma’s top leaders and professionals by taking part in this year’s Veeva R&D and Quality Summit. The Summit will allow GlobalVision to bring our automated quality control solutions directly to life sciences professionals and showcase our file inspection technology as a leading enterprise solution.
Currently, the pharmaceutical industry is being faced with a content efficiency problem that GlobalVision’s cutting-edge document comparison software can fix. At the Veeva R&D and Quality Summit, GlobalVision will showcase our platform and present our solutions, while networking and collaborating with pharma professionals to learn about the latest developments and best practices in life sciences.
Veeva R&D and Quality Summit
Venue: Hynes Convention Center, Boston, MA
Date: October 19-20th, 2022
The annual Veeva R&D and Quality Summit is bringing the industry back together in person this year in Boston. On October 19-20th, leaders and experts across clinical, quality and manufacturing, regulatory, safety, and IT will have the opportunity to connect face to face to learn about all the new innovations and progress that has been made in pharmaceuticals in the past year and more.
This two-day event dedicated to the life sciences will attract an exclusive audience of R&D professionals, life sciences decision-makers, influencers, and many more. Key themes that will be discussed this year include, breaking barriers to advance life sciences, transforming quality management across the value chain, and enabling safety as a strategic partner, just to name a few.
If you are interested in learning more about this year’s event, click here.
GlobalVision as a Veeva Partner
A limited number of partner sponsorships are offered by Veeva on an invitation-only basis. GlobalVision, a silver-certified Veeva technology partner, is very proud to announce they will be participating as a gold sponsor at the 2022 Veeva R&D and Quality Summit.
This past year, GlobalVision became a silver-status technology partner, and in doing so integrated our solutions with Veeva’s to help industry professionals solve their content efficiency problems.
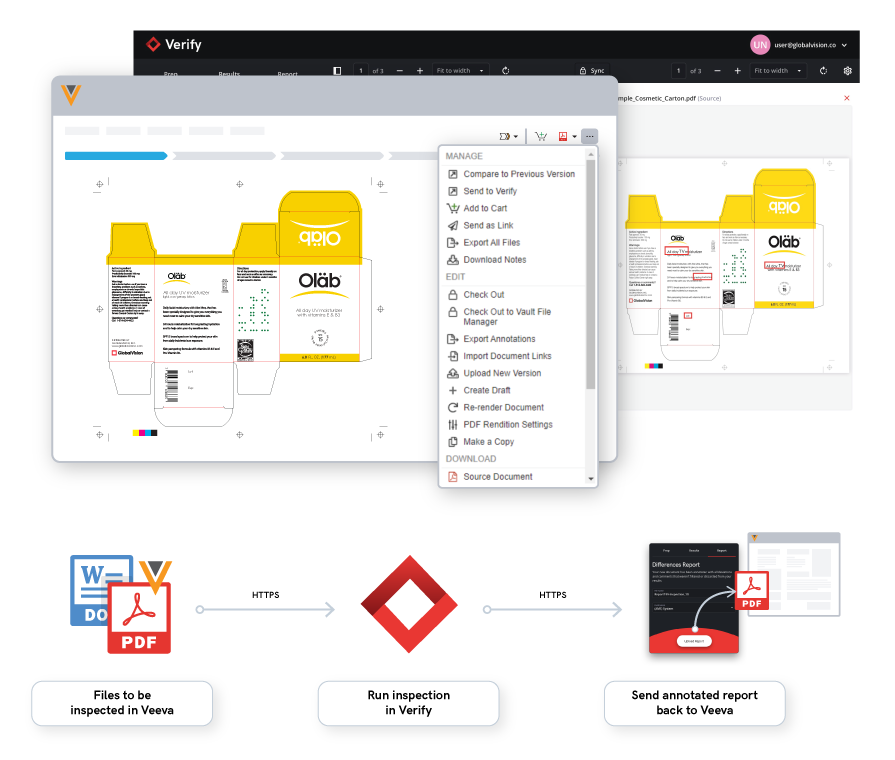
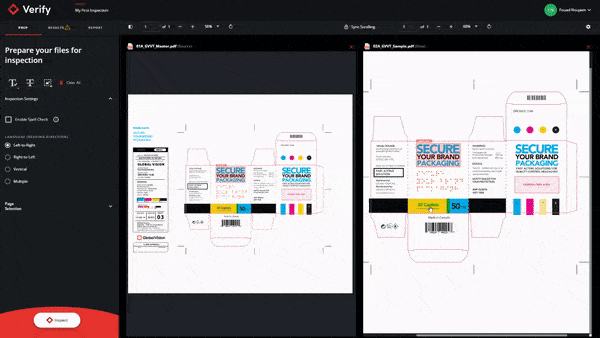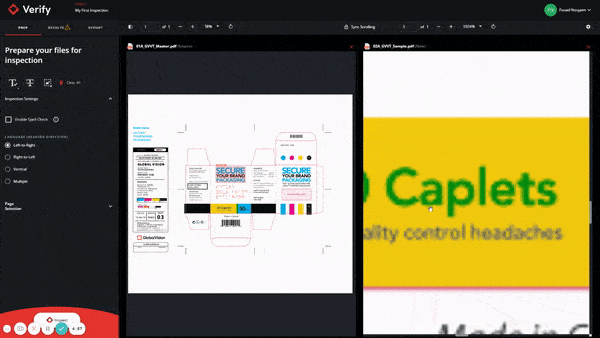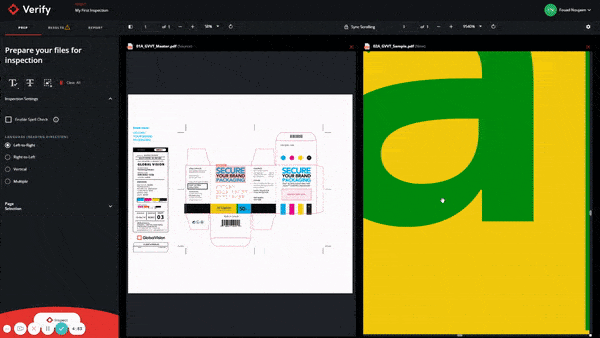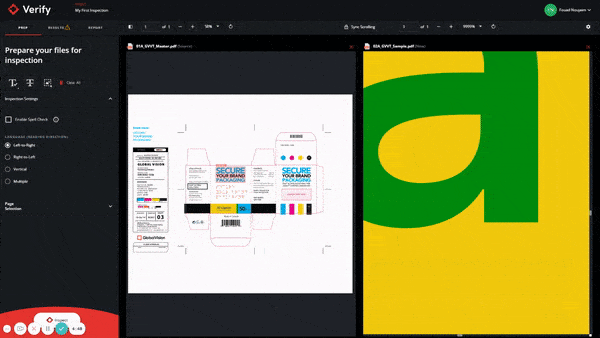
In short, GlobalVision’s cloud-based inspection application, Verify, has been integrated with Veeva Vault PromoMats and Veeva Vault RIM so users can speed up revision quality checks while ensuring any errors are quickly found. Any gaps and issues that may arise in the revision and inspection process have been solved as efficiency is now increased in the regulatory workflow.
Through the use of GlobalVision’s integration with Veeva Vault, this partnership has accelerated the document development and approval process tenfold and ensures only the highest quality content.
Participate in our Contest!
All attendees at this year’s event will have the opportunity to try their luck at the GlobalVision booth. Stop by to meet the team, learn about our products and automated solutions, and participate in our contest. All attendees need to do is fill out our GlobalVision card to enter for their chance to win valuable prizes. Prizes include the Apple Watch 8, Airpods Pro 2, a travel Theragun, and donations to a charity of choice.
If you are attending the Veeva R&D and Quality Summit this year, do not hesitate to drop by our booth to participate in our contest or reach out to a GlobalVision rep to set up an in-person demo.
We are excited to see you all live at this year’s event!
———————————————————————————————————————————————————
Related Resources:
- Find out more about Verify by visiting our website here.
- Find out more about Verify’s integration with Veeva.
- Veeva x Verify: Empowering Promotional, Regulatory, and Labeling Teams in Life Sciences to Maximize Content Efficiency
- Ready for a New Verify Release? 2023.2 is Here
Ensure your content is always error-free in record time with GlobalVision. Try it now for free.
Keep up with the latest updates in automated proofreading software. Sign up for our newsletter.