Date: April, 2019 | Category: Quality | Author: Mike Malz
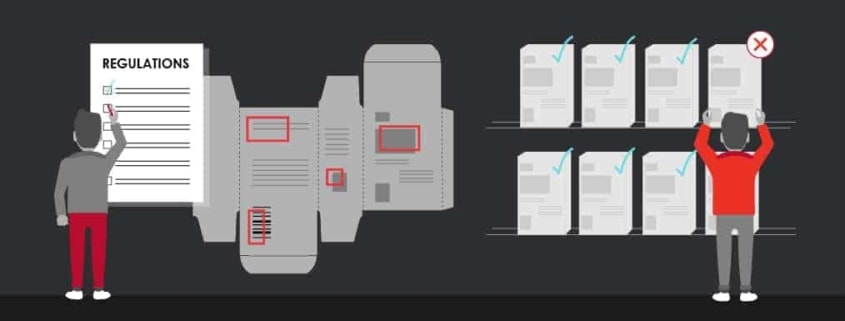
Regardless of the industry, all companies have the same goal in mind: to provide high-quality products to keep customers loyal and attract new ones. To accomplish this, each product must go through a strict proofreading process to ensure the accuracy of all labeling and packaging.
This can be time-consuming. As a result, some early adopters have switched to proofreading software due to time constraints. In fact, sticking with human proofreaders has proven to be a potentially costly mistake, one that may even lead to expensive recalls.
Avoid Manual Errors
Although they do their best, human proofreaders are not able to catch all errors that can be found on a label. In a North Carolina State University article about errors in food manufacturing, 25% to 50% of recalls were due to human error. There are many reasons why these errors were missed; For one, a proofreader could have been tired or simply unfocused.
With new technologies continuously introduced into every aspect of our lives, people can be distracted by an email alert or text message. When an error is detected in packaging, companies must issue a recall on their product, costing them thousands of dollars and leaving them with a ton of unsellable inventory. Fortunately, there are measures to ensure each packaging component is error-free. These measures can be combined into a single solution: an automated proofreading platform.
Proofreading That Checks Every Word
Human brains are trained to skip words. So, no matter how fluent they are in a language, they can overlook one that’s misspelled. Most people use spell-checking software such as the spellcheck feature in Microsoft Word or maybe even an online solution like Grammarly. These tools help catch those mistakes, but they do have flaws. For example, some English words are spelled differently, such as “color” and “colour.” One solution would be proofreading software that automatically detects errors in a loaded file.
Catch Discrepancies in Color
Despite evidence to the contrary, some may argue human proofreaders do catch misspelled words. Nevertheless, color differences are a different kind of animal. It’s important that the labels of packaging have a consistent color pattern. This helps promote brand awareness and identity. If the color has faded or the hue is slightly different, the product will come off as rushed. Color-detection software is one way to identify any issues before the file gets sent to the printing press.
A+ Barcode Grading
Every company wants to send out Grade “A” barcodes and nothing less, as recalls can be issued if they will not scan. And, yet, despite the very real need, human proofreaders are unable to tell if a barcode will scan properly when it reaches the customer. Leveraging technology is the only option here, and it makes sense to opt for the best solution possible. Having a process in place to digitally check barcodes saves you time and money. This is especially true when it comes to consumer goods.
Checking Labels Even After Printing
There is still a chance some errors or color differences will slip through the cracks even after the job is done and the labels are printed. The best way to review your printed files is by reverifying them, using proofreading software. This way the user can find any differences and decide to reprint if necessary.
It’s important to make sure that, even when the final label is produced, it remains error-free. It would be infinitely preferable to find errors in labels immediately after they’ve been printed instead of having a customer find them while in line at a store.
Making sure your products are high-quality extends to the packaging. Errors are not only costly but embarrassing. People are far from infallible, and companies that rely on human proofreaders risk having their products recalled and losing customers.
Making the switch to automated proofreading, like with GlobalVision, can save you time and money by sparing your firm from having to issue recalls. Businesses should consider spending a little more money now on solutions that help maintain their reputation and brand integrity. It’s not about short-term gains, but rather the long game, in which everyone is a winner.
_________________________________________________________________________________________
Get your complete guide to meeting FDA labeling requirements here
Learn how some of the world’s top pharma companies have cracked the efficiency code with automation