How to Ace FDA Food Label Compliance Review: Ensure Your Labels are Always Approved
Date: January, 2023 | Category: Compliance | Author: Hana Trokic
Every food item that makes it to the shelves of grocery stores worldwide undergoes rigorous testing and verification to ensure its safety.
To be precise, it is the responsibility of the Food and Drug Administration (FDA) to assure this guarantee and also to check that all foods are properly labeled and that they follow food label compliance.
This is necessary to give consumers the correct information about the food they buy and consume and to avoid any unnecessary adverse effects that may come with the improper labeling of said products.
From the ingredients list, allergens, date of packaging, and expiry date, all of this information is essential. Yet, more crucial than the information itself is the accuracy of the information on food labels and packaging.
The accuracy of their labels guarantees consumer safety and health, and also allows businesses to avoid recalls and substantial financial losses, while protecting their brand and public image.
In an industry that works in highly regulated and monitored conditions, even the slightest error can lead to enormous consequences. As such, food manufacturers and those working in consumer packaged goods cannot risk faulty labels going out to market.
To ace FDA food label compliance reviews and ensure your labels are always approved, tap into the power of automation. Automated quality control is a comprehensive solution for your compliance needs that ensures all of your food labels meet FDA label compliance review.
Innovative and accurate software proofreads critical content at scale and instantly catches errors to help you get your products to market faster without compromising on quality. You can now rest easy knowing your food labels are completely error-free all the while following FDA food label compliance and passing all reviews and tests.
Food Labeling at a Glance

Food labels have very defined rules regarding how they should be placed on packages and containers.
General Food Labeling Requirements
Generally speaking, there are two ways to label food:
- Place all required label statements on the front of the label panel.
- Place certain specified label statements on the principal display panel (PDP, or the part of a food label that is most likely to be displayed to the customer when for sale – the front label on a product) and other labeling on the information panel.
No matter the food product, the PDP must display specific information critical for consumers. This includes the food name and the net quantity statement. Food must also include the information panel, which is usually placed to the right of the PDP.
The information panel refers to statements that are generally required to be placed together. They usually include information such as the name and address of the manufacturer, ingredient list, nutrition labeling, and allergy labeling.
It is also important to note that label standardizations must be followed to ensure consistent and easy-to-read labels. The FDA requires that you use a prominent print size and that the font is clear and legible through specified letter height. Additional formatting is also detailed in FDA guidelines to ensure that text is easy to read on different backgrounds and artwork.
Name of Food
Every food label should be appropriately named so that consumers and the general public know exactly what the product is. The name of the food is referred to as the statement of identity and is required by the FDA to be on the front label.
As packaging for various yet very different food products may look the same, food products must be appropriately labeled to set them apart at first glance.
For a more detailed look at how the FDA breaks down naming food on labels, view their detailed food labeling guide here.
Net Quantity of Contents Statements
The net quantity statement provides information about the amount of food in the container or package. The FDA requires that it be expressed in weight, measure, or numeric count. If the food is solid, the contents are labeled in terms of weight. If it is liquid, they are labeled in fluid measure.
The net quantity statement must be placed at the bottom 30 percent of the PDP and must show net contents in both metric and U.S. Customary System.
Ingredients List
What is a food label without an ingredient list? One of the most crucial aspects of food labels and packaging, the FDA also clearly defines how ingredients should be written and displayed.
Firstly, ingredients are required to be listed in descending order of predominance. In other words, the most prominent ingredient (by weight) is listed first, and so on. The ingredient list is also placed on the same label as the name and address of the manufacturer.
Like other FDA food label rulings and regulations, the ingredients list is subject to strict standardization. Font and typography should be prominent and unified following the instructions given by the FDA, and the list should always be straightforward and easy to read.
Food Allergen Labeling
Under FDA label compliance, food labels are required to include all ingredients that are major allergens and can cause allergic reactions and potential harm to consumers.
While more than 160 foods have been shown to cause allergies to those with food sensitivities, only eight “major food allergens” must be labeled. This is because these ingredients, or an ingredient that contains protein derived from them, account for 90 percent of all food allergies.
The eight major food allergens include:
- Milk
- Egg
- Fish
- Crustacean shellfish
- Tree nuts
- Wheat
- Peanuts
- Soybeans
Nutrition Labeling
The nutrition label provides crucial information to consumers about the nutritional value of the product, broken down by calories, carbs, fat, fiber, protein, and vitamins per serving. It’s easily identifiable as the tabular, square, or rectangular-shaped information panel on all food products and packaging.
This label is usually displayed with the ingredient list and the name and address of the manufacturer. The location of the nutrition label is not precisely defined by the FDA and can be placed on packaging panels that can be clearly found and seen by consumers.
The FDA goes into great detail about nutrition labeling to ensure accurate information is being given and displayed to consumers.
For full details about FDA label compliance for nutrition labeling, see how the FDA breaks down this aspect of food labeling in their detailed guide.
Nutrition Content Claims
Nutrition content claims are claims made on food labels and packaging that imply certain nutrient characteristics of the food. This includes claims such as “low fat,” “high in protein,” and “only 100 calories per snack.”
We’ve all seen these claims on many food products and probably have several in our kitchen at any given moment. These claims aim to describe the level of certain nutrients in a specific food product.
They usually include words like free, high, low, reduced, lite, etc. The claims and labels are also regulated by the FDA but usually only apply to those with an established daily value.
Ace FDA Food Label Compliance Review Through Automation
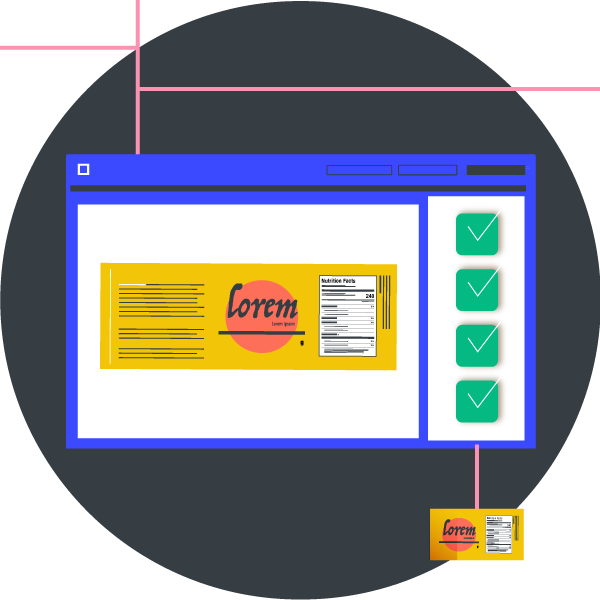 Food labels are critical. Not only do they provide vital information to consumers about the food choices they are making, but they are also legally and governmentally mandated and regulated. Those labels we see and read daily are crucial to ensuring the health and safety of the general public and consumer.
Food labels are critical. Not only do they provide vital information to consumers about the food choices they are making, but they are also legally and governmentally mandated and regulated. Those labels we see and read daily are crucial to ensuring the health and safety of the general public and consumer.
Suppose a typo or even a tiny error was to slip through. In that case, food manufacturers could face immense consequences, including large recalls, financial losses, and a negative brand image that could be difficult to reverse. The downfalls that come with faulty food labeling are often irreversible.
To save your company from going through horrific scenarios and prevent detrimental errors, food manufacturers should turn to automation as a foolproof solution to all proofreading needs.
Automated quality control makes proofreading processes more efficient and drastically eliminates the risk of errors slipping through in critical and highly regulated content.
In essence, automated quality control in proofreading is a file comparison tool that overlaps two files to find the discrepancies between them. No matter how minor the differences may be, the software will detect them, pinpoint their exact locations, and generate a comprehensive inspection report allowing users to review and navigate through all found differences.
A simple yet highly effective solution, all that is required from quality control teams is to upload both documents, conduct a scan, and view the discrepancies in the generated report.
By using automated solutions for their proofreading needs, food manufacturers can rest assured that their highly critical content is verified with 100 percent accuracy, in record time, and with absolutely no need for manual checks.
More than Just Text Inspection
While proofreading is often thought to be just for text, file comparison technology allows you to check, review, and inspect files wholistically. As food labels include different elements such as various fonts, typographies, graphics, and artwork, manufacturers can scan complete files in one go without having to do multiple inspections or reviews.
A file comparison tool can detect errors in the following:
Whatever discrepancies may appear in various revision cycles, they will all be detected within seconds to ensure a flawless master copy. GlobalVision’s file comparison software scans and inspects all files to find even the most minor errors allowing you to publish accurate and pixel-perfect labels.
Perfect Labels, Every Time.
Critical content in highly regulated industries should undergo rigorous and detailed reviews, but that doesn’t mean they have to be complicated and time-consuming. Automated quality control allows for thorough inspections that increase efficiency while decreasing review times and errors.
Food manufacturers can expect to experience countless benefits and advantages by implementing automated quality control – a leading proofreading solution for all regulated industries. An easy fix that ensures that all of your labels meet FDA food label compliance reviews, automated quality control is a comprehensive solution that does it all.
Enhance your proofreading practices and make the switch today. See how automated quality control can streamline your revision workflows by allowing you to proofread critical content at scale while maintaining complete accuracy.
Discover firsthand the benefits of automated quality control. Request a demo of GlobalVision, and get started today!
———————————————————————————————————————————————————
Related Articles:
- Solving The Content Efficiency Problem in the Pharma Industry
- Your Complete Guide to Meeting FDA Labeling Requirements
- Overcome Your Content Challenges in Cosmetic Labeling to Meet FDA Requirements
- Ensure Your Labels Meet all FDA Drug Labeling Requirements with Automated Quality Control
- How to Ease the Labeling Proofreading Process for Medical Devices
Ensure your content is always error-free in record time with GlobalVision. Try it now for free.
Keep up with the latest updates in automated proofreading software. Sign up for our newsletter.