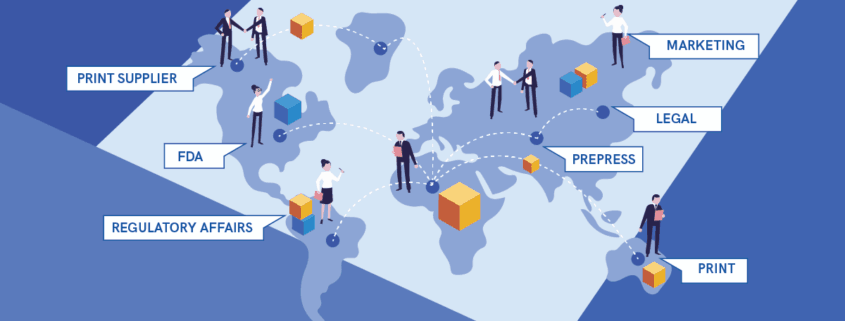
How Different Departments Help Develop and Proof Packaging
Date: May, 2019 | Category: Proofreading | Author: Ryan Szporer
It’s easy to imagine how much work goes into the production of packaging materials.
The Packaging Proofing Process
Even looking at the process in a vacuum, the back-and-forth alone in trying to finalize a design may spark images of heated boardroom debates on details as seemingly trivial as font size. However, before the packaging even gets to that point, it may shock you just how many steps are involved, in developing it from its infancy.
That’s not even taking into account factors like cost-effectiveness (including Return On Investment), time to market, and, perhaps most importantly, quality (accuracy). They all contribute to determining the degree of success of given packaging.
In effect, it’s a company-wide process of finalizing packaging (along with what’s inside), one that starts at the very top. Considering the financial ramifications of a mistake making its way onto the final product, it arguably stops there too, with one constant throughout the workflow being the need for thorough quality control.
Packaging Proofing in Pharma
Take a hypothetical company in the heavily regulated pharmaceutical industry, for example. Headquarters coordinates the launch of new drugs. And, while drugs themselves will likely be produced and packaged at different sites, the individual branding elements that go into the design are most probably determined at the head office. In effect, headquarters doesn’t just coordinate the supply chain, but all the different departments, including, in this case, marketing.
Input may come from the marketing units of offices around the world. It simply makes sense that they would get the chance to offer their two cents, especially with regard to packaging that will differ from one region to the next. Some of the content will nevertheless stay universal. Aside from the obvious elements (product name and branding), that content will take the form of drug and dosage information, for example.
In pharma, the Food and Drug Administration is the American regulatory body responsible, and documentation, like drug applications, has to be approved. A given firm’s Regulatory Affairs (RA) department liaises with the FDA, all the while reviewing packaging and labeling to ensure accuracy. It’s one of the first stages at which proofing becomes critical, to prevent typos and reduce back and forth. Truth be told, it’s just one of many product-development considerations with which RA helps, acting as a sort of bridge between departments.
The Reality of Conversion Errors
For example, with the approved copy (as well as technical and corporate-defined design requirements) in hand, the graphics or creative department begins to develop the file. The process is fairly intensive with a fair amount of artwork-creation checklist items for which to account, including making the file as accessible as possible to all other departments.
On that subject, even IT enters the fray, albeit indirectly. IT may not have any work to do with regard to the creation process, but the department is responsible for ensuring the homogeneity of software and platforms across the organization wherever possible.
The risk of conversion errors sneaking in at some point must be addressed. While it’s almost impossible to protect against the printer introducing them once the files get sent over, it’s relatively easy to ensure nothing of the sort occurs in-house simply by standardizing so that the files stay the same when being opened on different systems in each of the departments.
After all, to successfully determine whether the printer’s proof and subsequent shipment of packaging components match what had been approved internally, first, you have to make sure the file that had been approved internally actually appears as it should.
Approving Printed Packaging Materials
From there, it’s a matter of the internal quality control department proofing the shipment from the printer, ideally through proper sampling. The approved packaging then moves to production and gets distributed, ultimately landing in the hands of consumers. At that point, the packaging technically undergoes one final quality check.
Once the threat of expensive, required recalls or reprints dissipates, the quality objective can be safely confirmed to have been met. Assuming an efficient revision process, which can similarly be enhanced with the right digital proofing system, had been leveraged, so can the other two.
The trick is being able to sign off with confidence on the packaging before it even gets to that stage, though. It’s not so much a matter of bypassing the customer as it is of truly ensuring its quality for them and mitigating any risk ahead of time. Many different departments may be at work here, but proper proofing helps keep the operation running smoothly, limiting the number of revisions in play and ultimately the number of errors that slip through the cracks.
_________________________________________________________________________________________
Get your complete guide to meeting FDA labeling requirements here
Learn how some of the world’s top pharma companies have cracked the efficiency code with automation