Streamlining Pharmaceutical Artwork Management
Date: May, 2020 | Category: quality| Author: Luisa Berger
Developing pharmaceutical packaging can feel as complicated and lengthy of a process as developing the product itself.
If left without logical structure, this process quickly becomes vulnerable to inconsistencies and human limitations that can slow down workflows, cause unnecessary reworks, and put companies at risk of a recall. The good news: implementing standardized processes and leveraging automation can protect companies from these vulnerabilities and accelerate their existing workflows.
Pharmaceutical Artwork Management: A Non-Linear Process

The creation of a pharmaceutical label or package would seem to be a relatively linear process as the information moves from regulatory departments and core data sheets to designers and layouts and finally to printers and finished materials.
Yet, this relatively straightforward sequence quickly erodes with the countless revision and approval cycles that are introduced as a result of new upstream data or information, regional requirements, regulatory feedback, and many other influencing factors. The development and review of each of these stages can already be a lengthy process, making any additional errors or inconsistencies introduced along these workflows a potential bottleneck or implication further downstream.
Ideally, at every new version or hand-off, a comprehensive review and comparison of the current version to the previously approved one would take place. The goal is to ensure that by verifying content accuracy and correcting errors and inconsistencies at each stage, the entire process can be streamlined for more efficient and effective work, rather than frequent, easily avoidable reworks.
While, in theory, it sounds great to thoroughly inspect every project and every version like this, in reality, these reviews can take hours, even days when manually checking content that can range from less than 12 words to over 120 pages. Enter automation.
Automation for Packaging Artwork Management
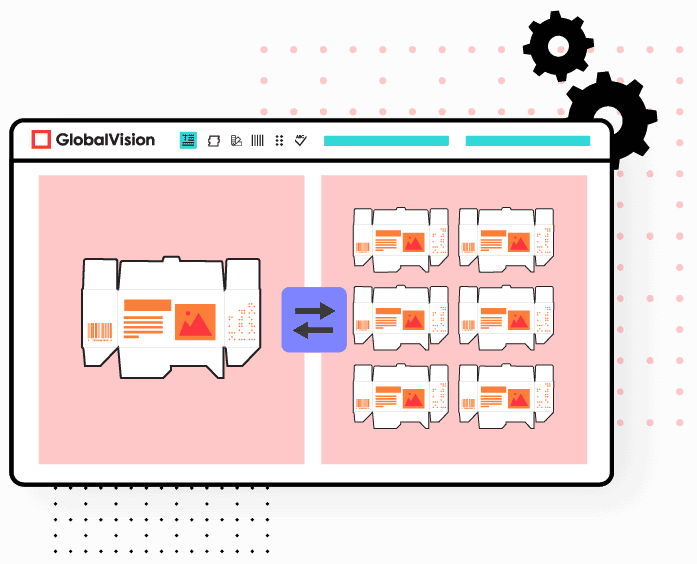
Automated inspection technologies are to these processes what a calculator is to engineering: a tool that quickly, reliably, and with precision performs functions that humans can do, but with significantly more time and greater room for error.
Automation acts as an aid, not a replacement, for human participation. Rather, by allowing computers to do what they do best, teams are actually able to apply their attention and efforts to those actions that, alongside these automated processes, keep projects moving forward and time-to-market speeding up.
From original source documents and manuscripts to design layouts and graphics to physical proofs and finished materials, automation can quickly and consistently verify the diverse components and elements that make up these projects. With text verification tools, you can ensure that your copy was properly transcribed or conserved across versions and formats, fool-proofing that no errors or inconsistencies slipped through review.
Graphics errors, like missing logos, inaccurate graph drawings, or color inconsistencies, can be instantly highlighted using pixel overlay technologies. Even verification processes that already benefit from some innovation and standardization, like barcode and braille authentication and translation, can be further optimized using automation for faster, more informative results. Automated solutions like these are readily available tools that can be easily leveraged to transform productivity and optimize outputs.
Bridging the Gaps in Artwork Management

While these tools and capabilities can transform traditional processes and manual verification methods, when integrated into existing product lifecycle management (PLM) systems, these solutions can bridge the existing quality gaps both internally, within and across departments, and externally with vendors.
In today’s context, most pharmaceutical and life science companies utilize software and digital management systems for organizing projects, document management, and other workflows. By seamlessly integrating into these existing platforms, these routine quality checks become an easily accessible and implementable procedure for verifying each contribution that benefits the efficiency and collaboration of everyone participating in that workflow.
Given that companies in this space outsource those functions that are not within their necessary expertise, whether marketing, printing, or other vendor services, quality standards must also apply to this level. When both company and vendor employ the same quality control tools, there is a common language and agreed-upon standard that both parties are committed to upholding and remaining accountable to.
Beyond Workflows

While these tools are optimizing the artwork development and review process for faster, more streamlined projects, these integrations also allow businesses to collect relevant data on areas of their workflows, like the number of revisions, common errors, vendor performance, and other relevant metrics. Ultimately, these insights can help inform further strategies and business efforts for optimization and improvement in these areas and beyond.
Unlike desktop-only or shared, physical verification tools, cloud-based solutions allow teams to work independently and securely wherever they can access an internet browser. Whether integrating with existing systems or opting for a cloud-based solution, companies can ensure that their team’s ability to access their quality control tools and uphold standards is disruption-proof.
With more companies supporting working remotely, or external conditions limiting access to workplaces or tools, these easily accessible solutions allow teams to continue thriving and delivering flawless results, regardless of the circumstances.
The artwork management and creation process is an extensive undertaking that involves wide-ranging collaborative efforts across and beyond pharmaceutical companies. By implementing coherent processes and leveraging automated solutions, these non-linear workflows can become more streamlined for faster outputs and fewer errors slipping through the cracks.
Curious about what effects this kind of automation could have on your own workflow? Book a process consultation with one of our representatives to explore how automation could work for you.
_________________________________________________________________________________________
Ensure your content is always error-free in record time with GlobalVision. Try it now for free.
Keep up with the latest updates in automated quality control. Sign up for our newsletter.
Get your Complete Guide to Meeting FDA Labeling Requirements Here
Learn How Some of the World’s top Pharma Companies have Cracked the Efficiency Code with Automation